Duck-Billed Platypus Venom: Designed for Discovery
I wouldn’t classify it as a bucket-list experience, but it was off-the-charts cool to see a duck-billed platypus up close a few years ago when my wife and I visited Tasmania. This little creature reminded me of a beaver as he swam around in the water.
But as cute and cuddly as the duck-billed platypus appears to be, I came to learn (not by experience but by listening to the zookeeper) that you don’t want to mess with this egg-laying mammal. The platypus has spurs on its hind feet, and for males, the spurs are loaded with venom. Being struck by a platypus’s spurs is no pleasant thing. The venom can kill a small animal (such as a dog) and cause excruciating pain for humans.
Not only does the duck-billed platypus fascinate animal lovers, it has captured the attention of the scientific community. This creature is neither a placental nor a marsupial mammal. Instead, it belongs to an unusual group called the monotremes. Biologists regard monotremes as primitive mammals. And because they group apart from other mammals, many life scientists believe that they can learn a lot about the mammalian biology (including human biology) through comparative studies of the monotremes.
Recently, researchers from Australia demonstrated the value of studying platypus biology when they discovered that a gut hormone (GLP-1) which regulates blood sugar levels doubles as a component in the duck-billed platypus’s venom.1 They believe that this insight may lead to a new drug treatment for type 2 diabetes.
To appreciate why this research team thinks that the platypus GLP-1 hormone may have use in treating diabetes, a little background is in order.
GLP-1
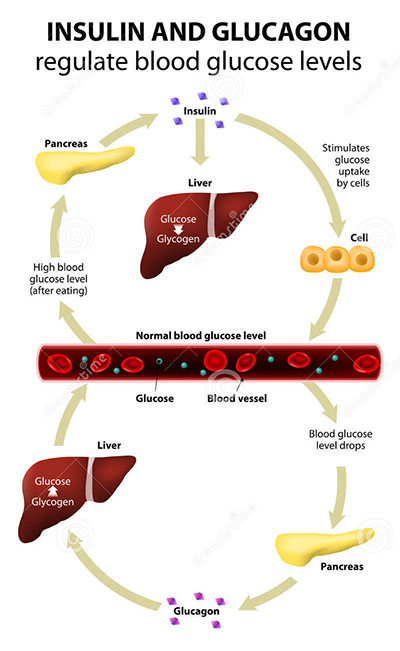
Found in all mammals, glucagon-like peptide-1 (GLP-1) belongs to a family of biomolecules called incretins. These compounds serve as metabolic hormones that stimulate a decrease in blood glucose levels. Secreted in the gut, GLP-1 ultimately lowers blood sugar levels by making its way through the blood stream to the pancreas. GLP-1 stimulates the beta-cells in the pancreas to release insulin. In turn, insulin causes the liver, muscles, and adipose tissues to take up glucose from the blood.
GLP-1 is named after glucagon. A blood hormone, glucagon has the opposite effect as insulin. When released by the alpha-cells of the pancreas, glucagon stimulates the liver to break down glycogen and then release glucose into the blood stream. Glucagon exerts its effect when the blood sugar level drops. Like GLP-1, glucagon also stimulates insulin release, so when the blood sugar level rises (because of glucagon’s release from the alpha-cells), the sugar is quickly taken up by muscle and adipose tissues.
Image: Insulin and glucagon regulate blood glucose levels in the human anatomy, specifically the liver and pancreas.
Eating food stimulates the release of GLP-1 in the gut. This ingenious design ensures that insulin is released and the liver, muscles, and fat tissues are poised to take up glucose even before blood sugar levels rise as nutrients are absorbed into the bloodstream via the digestion process. This preparation is vital, because elevated levels of blood sugar have dangerous long-term consequences.
Platypus Venom
To the surprise of the Australian researchers, the venom of the duck-billed platypus contains GLP-1. Other animals, such as the Gila monster, have venom components that are structurally analogous to GLP-1, but are distinct molecules. (In the Gila monster, this bio-compound is called exendin-4.) Again, an ingenious design. Including incretins in venom causes blood sugar levels to drop after the venom is injected into the victim. Lowered blood sugar levels create confusion and lethargy.
Unlike GLP-1, GLP-1-like venom components of, say, the Gila monster, are long-lived in the bloodstream because they have structural features that make them resistant to digestive enzymes such as dipeptidyl peptidase. This enzyme targets GLP-1 after its release to ensure it is quickly destroyed once this gut hormone triggers insulin release. If not quickly removed, insulin release would persist, thereby causing blood sugar to plummet to dangerously low levels.
The structure of the GLP-1 produced by the duck-billed platypus appears to be fine-tuned so that this biomolecule can balance its two roles as a gut hormone and a venom component. And this property makes the platypus GLP-1 an intriguing molecule to biomedical scientists looking for more effective ways to treat type 2 diabetes. The duck-billed platypus GLP-1 is an actual gut hormone (as opposed to an analog), but is much longer lasting, which makes it an ideal anti-diabetic drug.
Type 2 Diabetes
The most common form of the disease, type 2 diabetes results primarily from lifestyle effects: namely, obesity and lack of exercise. (Although there also appears to be a genetic contribution to this form of diabetes.) In type 2 diabetes, the capacity of beta-cells in the pancreas to secrete insulin becomes impaired, usually because of the accumulation of amylose in their interior. Reduced insulin secretion causes blood sugar levels to remain elevated at dangerously high levels. Persistently elevated blood sugar levels can lead to heart disease, stroke, loss of vision, kidney failure, and impaired blood circulation to the extremities.
Treatment for type 2 diabetes centers around dietary changes designed for keeping blood sugar levels low, weight loss, and increased exercise. Anti-diabetic medications also play an important role in managing type 2 diabetes. Pharmacologists have developed an arsenal of drugs, but all of them have their shortcomings.
Platypus GLP-1 as an Anti-Diabetic Medication
Because of the limitations of current anti-diabetic medications, pharmacologists are intrigued by the platypus version of GLP-1. Like all variants found among mammals, this gut hormone lowers blood glucose levels, but because it doubles as a venom component, it has a longer half-life than the GLP-1 hormones produced by other mammals—an ideal set of properties for an anti-diabetic drug. In fact, there is already a precedent for using venom components to treat diabetes. Exendin-4 from the Gila monster has been developed into a last resort anti-diabetic drug called Exenatide.
The Case for Evolution, the Case for Creation
It’s provocative that the biology of a creature, such as the duck-billed platypus, could provide such important insight into human biology that it can drive new drug development, positively impacting human health.
This study highlights the clever designs that characterize biochemical systems. The function of GLP-1 as an incretin and, in turn, its employ as a venom component are nothing less than genius. The elegance and sophistication of biochemical systems are precisely the characteristics I, a Christian biochemist, would expect to see, if, indeed, life stems from a Creator’s handiwork. In contrast, sophistication and ingenuity aren’t the features I would expect if evolutionary mechanisms—which are unguided, co-opting preexisting designs and cobbling them together to produce new designs—have generated biochemical systems.
Still, many people in the scientific community would argue that as hard as it may be to believe that biochemical systems evolved, it must be the case. Why? Because of the shared features that characterize these systems. As a case in point, the GLP-1 gut hormone is found in all mammals. So, presumably, this biomolecule emerged in the evolutionary ancestor of mammals and persists in all mammals today. Likewise, the shared features of GLP-1 and exendin-4 found in the Gila monster venom indicate to many biologists that the venom component must be evolutionarily derived from GLP-1.
Yet, as a creationist and an intelligent design proponent, I choose to interpret the universal nature of the cell’s chemistry and shared features of biochemical systems as manifestations of archetypical designs that emanate from the Creator’s mind—inspired by the thinking of Sir Richard Owen. To put it differently, for me, the shared features reflect common design, not common descent.
Of course, this leads to the follow-up rebuttal: Why would God create using the same template? Why not create each biochemical system from scratch to be ideally suited for its function? As I pointed out recently, there may well be several reasons why a Creator would design living systems around a common set of templates. In my estimation, the most significant reason is discoverability. The shared features of biochemical systems make it possible to apply what we learn by studying one organisms to all others, in some cases. As a case in point: The occurrence of GLP-1 in all mammals and the shared features of GLP-1 and exendin-4 make it possible to gain insight into human biology by studying the duck-billed platypus.
This discoverability makes it easier to appreciate God’s glory and grandeur, as evinced in biochemical systems by their elegance, sophistication, and ingenuity.
Discoverability of biochemical systems also reflect God’s providence and care for humanity. If not for the shared features, it would be nearly impossible for us to learn enough about the living realm for our benefit. Where would biomedical science be without the ability to learn fundamental aspects about our biology by studying model organisms such as yeast, fruit flies, and mice? How would it be possible to identify new medications if not for the biochemical similarities between humans and other creatures, such as the duck-billed platypus?
Far from making no sense, the shared features in biochemistry are a manifestation of the Creator’s care and love for humanity.
Resources
- “Archetype or Ancestor? Sir Richard Owen and the Case for Design” by Fazale Rana (article)
- “Q&A: Why Would an Infinite Creator Employ the Same Designs?” by Fazale Rana (article)
Endnotes
- Enkhjargal Tsend-Ayush et al., “Monotreme Glucagon-Like Peptide-1 in Venom and Gut: One Gene—Two Very Different Functions,” Scientific Reports 6 (November 29, 2016): id. 37744, doi:10.1038/srep37744.